| 2015 | APR | |||||||
| 2014 | JUL | OCT | DEC | |||||
| 2013 | JAN | FEB | APR | MAY | JUN | JUL | AUG | SEP |
| 2012 | SEP | OCT | NOV | DEC |
Chondroitin sulfate - the mucopolysaccharide
About chondroitin sulfate
Chondroitin sulfate falls into the category of mucopolysaccharides, the saccharide compounds related to starch. Commercially available chondroitin sulfate comes in the form of a clear white powder that very much resembles white salt. The big difference between mucopolysaccharides and starch is that starch has a relatively simple structure. It does not contain nitrogen and has no antigenicity, but mucopolysaccharides have a more complicated structure with a nitrogen atom inside its molecules, and it often possesses antigenic properties. Proteins and nucleic acid are the important compounds that contain nitrogen and are well known for their immunogenic (antigenic) qualities, but the immunogenicity of mucopolisaccharides is in no way inferior to these substances. In other words, mucopolysaccharides such as chondroitin sulfate, not unlike proteins, are sustaining our existence right this moment! Mucopolysaccharide is actually the old name for proteoglycan, but it is still being used by many researchers. If we were to summarize the properties of chondroitin sulfate, we should say that all its physiological and pharmacological characteristics are defined by its chemical structure—it is a large compound molecule with a strong negative charge. Besides chondroitin, there are other mucopolysaccharides inside of our bodies that are parts of protein molecules and all work synergistically.
About sugars
Commonly associated with sweets, chemically, sugars are carbon compounds with a hydroxyl group and a reduced group contained within one molecule. The sugar with the simplest structure is called monosaccharide, and its common representative, glucose, is present in the human body and in most animals and plants. The amount of glucose produced by plants on Earth is said to total more than one billion tons annually. When two monosaccharides are connected, it is called disaccharide, and when there are three, it is called trisaccharide. The best-known disaccharide is called saccharose or table sugar—this is the sugar that we commonly use in cooking. Saccharides made of 2-10 molecules of monosaccharide are categorized as oligosaccharides, and those made of 11 or more molecules are called polysaccharides.
Monosaccharides
Grape sugar (glucose) This is the most frequently produced monosaccharide on Earth. Its sweetness is about 70% that of saccharose, and it is contained in human blood (about 0.1%). It is contained in many fruits, such as peaches (4%) and pears (4%).
Fructose is contained in sweet fruit; for example, its content in grapes, apples, and pears is about 5-8%. This saccharide is known as the sweetest (about 1.7 times sweeter than saccharose). It is contained inside the human body (about 0.5%).
Galactose Its sweetness is about 30% that of saccharose. A lot of galactose is found inside the nerve tissue of the brain. Inside the human body, it is synthesized from glucose and is not found in a free form.
Oligosaccharides
Saccharose (sucrose). This is the commonly known table sugar used in cooking. Chemically, it is a disaccharide, and until it is enzymatically decomposed into monosaccharides, it cannot be absorbed in the human intestine.
Lactose This disaccharide is synthesized inside the mammary gland and contained only in milk. Its sweetness is about 16% that of sucrose.
Maltose This is an ingredient in many sweets, and it is produced by decomposing starch with a malt solution. It is not commonly found in nature.
Fructooligosaccharide (oligofructose) This saccharide is made of sucrose and fructose chained in one or more links. It is found in such plants as burdock and onion, and although it is not absorbed in the human digestive system, it is consumed by Lactobacillus bifidus bacteria inside the large intestine and therefore considered health giving.
Polysaccharides
Starch is a polysaccharide made of multiple glucose links; it is produced by multiple plants and has become the main energy source for humans.
Glycogen This is a storage polysaccharide found in animals. It resembles amylopectin but has more ramifications. Its molecule is big (about several million), and it is found in the liver and joint cells in large amounts.
Cellulose is the most frequently produced carbon compound on Earth. Its annual plant synthesis amount is more than one trillion tons. Similar to starch, it is made of glucose molecules, but they are connected differently, making cellulose useless as the energy source for humans. Nevertheless, cellulose can become food for other animals (i.e., sheep, camels, etc.) that host microorganisms producing catabolic enzymes capable of splitting cellulose.
Chondroitin sulfate is also a polysaccharide; it is a typical mucopolysaccharide and was first separated from cartilaginous tissue in 1861. 90 years after the discovery of chondroitin sulfate, the three types of chondroitin, A, B, and C (the D type has been discovered recently) were discovered, which became possible largely because of enzymes (capable of splitting chondroitin sulfate into disaccharides) found by professor Sakaru Suzuki at Nagoya University.
Animal cartilage (such as chicken cockscomb, fish cartilage) has become the main source of chondroitin sulfate. Japan is the world leader in chondroitin sulfate production. In 1993, the total world chondroitin sulfate production was about 130 tons, 100 tons of which were manufactured in Japan. Out of these 100 tons, almost 90 were used in the medicine and food industries, and 10 tons were used in cosmetics.
 The word “chondroitin” was derived from Greek roots, and it means something similar to “the source of cartilage.” Cartilage tissue has unique qualities that are largely defined by chondroitin sulfate, its main component. Its complicated structure makes chemical synthesis inapplicable, so extraction from animal cartilaginous tissue and microorganism culture are the only options for industrial production. As multiple studies have revealed, chondroitin sulfate has a much wider tissue distribution inside the human body (compared to other mucopolysaccharides), being present not only in cartilage, bones, and other hard tissues but also in the brain and most other tissues and organs. It was also discovered that chondroitin sulfate is in charge of many vital functions of the internal organs. There are four types of chondroitin sulfate: A, B, C, and D. Among these, the only difference between A and C is the position of the sulfate group, which explains why these two have almost the same properties and why their distribution area in tissues is almost identical. By comparison, the B and D types vary in their properties and distribution.
The word “chondroitin” was derived from Greek roots, and it means something similar to “the source of cartilage.” Cartilage tissue has unique qualities that are largely defined by chondroitin sulfate, its main component. Its complicated structure makes chemical synthesis inapplicable, so extraction from animal cartilaginous tissue and microorganism culture are the only options for industrial production. As multiple studies have revealed, chondroitin sulfate has a much wider tissue distribution inside the human body (compared to other mucopolysaccharides), being present not only in cartilage, bones, and other hard tissues but also in the brain and most other tissues and organs. It was also discovered that chondroitin sulfate is in charge of many vital functions of the internal organs. There are four types of chondroitin sulfate: A, B, C, and D. Among these, the only difference between A and C is the position of the sulfate group, which explains why these two have almost the same properties and why their distribution area in tissues is almost identical. By comparison, the B and D types vary in their properties and distribution.
- Chondroitin sulfate A and C These types of chondroitin sulfate comprise the base of the cartilage and are of course contained in the connective tissues of vertebrates and other lower animals such as annelids, arthropods, mollusks, etc. Chondroitin sulfate in vivo is usually bonded with other mucopolysaccharides and proteins and is usually in the form of the so-called proteoglycan.
- Chondroitin sulfate B The structure of chondroitin sulfate B is similar to that of chondroitin sulfate A, and because it was first found in skin tissue, it is commonly known as dermatan sulfate. This mucopolysaccharide contains a rare saccharide, called idose, in the form of iduronic acid, which explains why its properties are different from those of other chondroitin sulfate types. Besides the skin, it is distributed in the lungs, heart valves, umbilical cord, and other tissues. Bound with collagen, it forms large molecules that are known to suppress the adherence of cells. This effect is especially important because it blocks the spread of cancer cells.
- Chondroitin sulfate D type was discovered in shark cartilage. Structurally, it resembles chondroitin sulfate C, but it has more sulfate groups, which explains its unique physiological effects.
Physiological effects of chondroitin sulfate
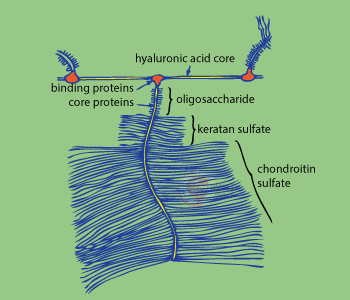
Mucopolysaccharides, which include chondroitin sulfate, are known to function while bonded with proteins in most of their forms. This makes such mucopolysaccharides as hyaluronic acid exceptions. The ratio of polysaccharides to proteins varies depending on the particular tissue. As shown in the picture, inside the proteoglycan contained in the cartilage, the incredibly long hyaluronic acid molecules serve as its core and protein molecules, which are also incredibly large (the molecular weight is as high as and 300,000 g/mole), are connected to it. With chondroitin sulfate and keratan sulfate also attached to this core, this so-called proteoglycan looks like a brush. It is still not clear why proteoglycans have such a shape, but it can be said for sure that, when a hundred of such proteoglycans are fortified by binding proteins and hang down from the hyaluronic acid cores, with the molecular weight of such a construction totaling more than 50,000,000 g/mole pathogenic bacteria that might be in its way are doomed.
Chondroitin sulfate is in charge of “logistics management” in our bodies.
Between most cells in the human organism is proteoglycan (containing chondroitin sulfate) that is always busy storing water, inorganic ions, vitamins, hormones, etc., and delivering them to cell acceptors when needed. Besides “logistics management,” proteoglycan also acts as a “customs office,” similar to glial cells (which help to form our nervous system), inspecting incoming particles and either accepting or rejecting their passage.
Chondroitin sulfate and water retention
Water is without a doubt one of the most important body constituents in humans (and other creatures). Proteins, which are often called the source of life, and chondroitin sulfate itself can perform their functions only when water is available as a medium. It is true to say that, at least for our solar system, that without water, there is no life. As shown in the table below, the water ratio for an average male is about 60%, for a female, it is about 51%, and for a newborn, it is almost 80%! There is a difference between men and women because women naturally tend to have more fat than men. Interestingly, the water content in muscles (about 75%), which look very firm, is not much different from that of the blood (80%) or the brain (68-84%).
|
Tissues and organs |
Water content (%) |
|
Human body |
|
|
Newborn |
79 |
|
Adult male |
60 |
|
Adult female |
51 |
|
Blood |
80 |
|
Brain |
68-84 |
|
Lungs |
76 |
|
Muscles |
75 |
|
Bones |
10 |
Water balance is essential for our well-being. A lack and an excess of water are both undesirable. The worst scenario is when water cannot be supplied. If the water content drops to only 10% below the norm, body cells can no longer perform their metabolic exchange. If the water content drops by 20% (for example, because of a cholera infection), it will surely mean death. To prevent a water shortage, an area deep in the brain (the hypothalamus) functions to control the water content throughout the body. This is possible because of the chondroitin sulfate contained inside proteoglycan, which itself is found in the inter-cellular substance of tissues and organs. Unfortunately, chondroitin sulfate can only store water. The opposite function (dehydration) is achieved with the albumin in the blood. Inside proteoglycan’s massive molecules, the acidic groups (i.e., sulfates) of the chondroitin sulfate and carboxyl groups are responsible for attracting water. These possess a negative charge and therefore can attract water molecules, but even stronger is their ability to pull sodium ions (Na+) and attract even more water through the osmolar phenomenon. Chondroitin sulfate’s ability to attract water is essential for the health of the skin.
Check out similar products here


