| 2015 | APR | |||||||
| 2014 | JUL | OCT | DEC | |||||
| 2013 | JAN | FEB | APR | MAY | JUN | JUL | AUG | SEP |
| 2012 | SEP | OCT | NOV | DEC |
Mechanisms of fucoidan’s anticancer action: 1, 2, 3 of 5
 In 1983, Takahashi (1), using crude fucoidan obtained from arame, investigated the effects on reticuloendothelial system phagocytic capacity, virus-inhibiting factor induction, and natural killer activity, effects on mice inoculated with the L1210 vaccine, and antitumor properties with respect to sarcoma 180 tumor cells transplanted subcutaneously in ddY mice and athymic nude mice. The results indicated that, by stimulating the reticuloendothelial system and increasing NK activity, arame brought about the emergence of antitumor properties while at the same time participating in T cell immunity. In 1996, researchers determined the chemical structure of U-fucoidan etc. and also brought clarity regarding their physiological activity, i.e., apoptotic effects on cancer cells. In 2000, Maruyama et al. (2) investigated the effects of wakame-derived fucoidan on NK cell activity in order to clarify the mechanism of life-extending effects exhibited by the substance on mice transplanted with L1210 leukemia cells. They administered water-soluble mekabu extract and fucoidan extracted from defatted, dried mekabu to mice intra-abdominally once per day consecutively at a dosage of 50 mg/kg body weight, and they confirmed the presence of NK cell activity-enhancing effects in the spleen and in cells exuded intra-abdominally after five days. Research by Sagawa et al. (3), Tominaga et al., Wu et al., Kato et al., and others has demonstrated bioactivity with regard to gagome-derived fucoidan in particular while also beginning to clarify the mechanisms of the anticancer effects. Moreover, anti-cancer mechanisms were reported on by Sakai et al. and Kato et al. (4). We will give an account based on these reports.
In 1983, Takahashi (1), using crude fucoidan obtained from arame, investigated the effects on reticuloendothelial system phagocytic capacity, virus-inhibiting factor induction, and natural killer activity, effects on mice inoculated with the L1210 vaccine, and antitumor properties with respect to sarcoma 180 tumor cells transplanted subcutaneously in ddY mice and athymic nude mice. The results indicated that, by stimulating the reticuloendothelial system and increasing NK activity, arame brought about the emergence of antitumor properties while at the same time participating in T cell immunity. In 1996, researchers determined the chemical structure of U-fucoidan etc. and also brought clarity regarding their physiological activity, i.e., apoptotic effects on cancer cells. In 2000, Maruyama et al. (2) investigated the effects of wakame-derived fucoidan on NK cell activity in order to clarify the mechanism of life-extending effects exhibited by the substance on mice transplanted with L1210 leukemia cells. They administered water-soluble mekabu extract and fucoidan extracted from defatted, dried mekabu to mice intra-abdominally once per day consecutively at a dosage of 50 mg/kg body weight, and they confirmed the presence of NK cell activity-enhancing effects in the spleen and in cells exuded intra-abdominally after five days. Research by Sagawa et al. (3), Tominaga et al., Wu et al., Kato et al., and others has demonstrated bioactivity with regard to gagome-derived fucoidan in particular while also beginning to clarify the mechanisms of the anticancer effects. Moreover, anti-cancer mechanisms were reported on by Sakai et al. and Kato et al. (4). We will give an account based on these reports.
-
Apoptosis induction.
One of the anticancer effects of fucoidan operates by driving cancer cells to kill themselves, that is, through apoptosis induction. The cells of living things include genes that command them to kill themselves when they have undergone aging because of an abnormal environment, and the death by suicide that occurs in this way is known as "apoptosis". Apoptosis enables the regenerative process whereby new cells are born when old cells die within the body to be repeated, maintaining physical health. This phenomenon of apoptosis occurs in humans as well. As a common example, although a newborn baby has fingers, they did not grow out when it was a fetus but rather formed at first in the shape of mits, after which only a certain number of certain cells undergo apoptosis at certain times, resulting in the formation of the fingers. Fucoidan is known to have effects acting directly on cancer cells that induce suicide. There are no side effects as this does not work on normal cells.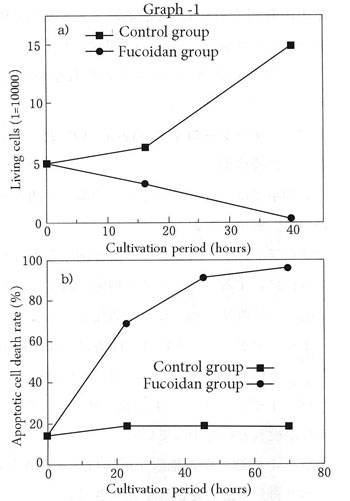 This apoptotic effect was shown in an artificial laboratory environment as a result of experiments by Yu et al. (5) investigating the effects of U-fucoidan on cancer cells that had been allowed to proliferate for many generations. Specifically, when they added U-fucoidan to human promyelocytic leukemia cells, human acute lymphoblastic leukemia cells, human stomach cancer cells, and colon adenocarcinoma cells that were actively proliferating in a petri dish, they found that the viable cell count decreased and approached zero, as shown in (a) in fig-1. This figure relates to the human promyelocytic leukemia cells. When they investigated in more detail they were able to confirm that in those cell deaths, the DNA that forms the blueprint of life was cut, and the cells became non-viable as in (b) in fig-1. They found almost no effect from U-fucoidan in normal cells that were cultured simultaneously as a control group.
This apoptotic effect was shown in an artificial laboratory environment as a result of experiments by Yu et al. (5) investigating the effects of U-fucoidan on cancer cells that had been allowed to proliferate for many generations. Specifically, when they added U-fucoidan to human promyelocytic leukemia cells, human acute lymphoblastic leukemia cells, human stomach cancer cells, and colon adenocarcinoma cells that were actively proliferating in a petri dish, they found that the viable cell count decreased and approached zero, as shown in (a) in fig-1. This figure relates to the human promyelocytic leukemia cells. When they investigated in more detail they were able to confirm that in those cell deaths, the DNA that forms the blueprint of life was cut, and the cells became non-viable as in (b) in fig-1. They found almost no effect from U-fucoidan in normal cells that were cultured simultaneously as a control group. -
Induction of interleukin 12 (IL-12) and interferon-gamma (IFN-γ) production.
In this section especially we need to use some rather obscure terminology, so I will give some explanations.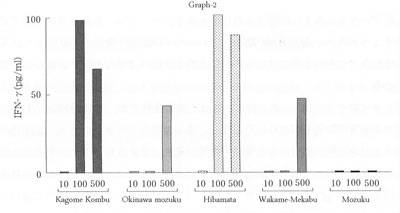
Macrophages: Also called phagocytes, these cells react when foreign substances enter the body. They take in viruses and bacteria and useless substances such as old cells into their own cells and eat and digest them. They eat the likes of cancer cells as well, one after the other. When there are a lot of enemies, they send out attack commands to their friends.
T lymphocytes (T cells): These cells go to work after receiving attack commands from macrophages. They include helper T cells, killer T cells, and suppressor T cells, each of which takes on a different role.
Killer T cells: Upon finding a cancer cell, this type of cell makes a hole in the cell membrane of the cancer cell and delivers a protein-degrading enzyme, destroying the cancer cell.
Helper T cells: Although they themselves do not attack cancer cells, helper T cells release cytokines such as interleukin-2, stimulating nearby macrophages and T cells in order to boost their offensive capabilities.
Suppressor T cells: These cells give commands to stop attacking when cancer cells have disappeared in the surrounding area due to macrophages and killer T cells.
Natural killer cells (NK cells): Independent of commands from helper T cells, natural killer cells go on the offensive immediately upon finding cancer cells or other foreign substances, destroying and eliminating cancer cells.
Interleukins (IL): Some types of cells secrete these bioactive protein substances externally under certain conditions. In many cases, the same kind of substances are commonly produced and secreted from cells other than lymphocytes as well, including macrophages. The general term for these substances is cytokines. Known interleukins include IL-1 through IL-18.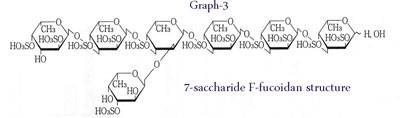
Interferon gamma (IFN-γ): An antiviral substance produced by lymphocytes. Produced by the above-mentioned T cells and NK cells, this multifunctional factor possesses many types of physiological activity, including antiviral activity, cell growth inhibition effects, antitumor effects, and macrophage and NK cell activation effects. Production of IL-12 and IFN-γ is known to be induced by fucoidan.
The example of induction by fucoidan of the aforementioned IL-12 and IFN-γ is as follows, according to the research of Kato et al. (4). In this case the researchers added gagome-derived fucoidan to the spleen lymphocytes of mice immunized against Meth A tumor cells, as shown in fig.2. When they added 1 - 100 μg/ml, they learned that, within that range, the more they added, the more was produced. However, when they looked at IFN-γ production amounts for fucoidan obtained from five kinds of brown algae, they found that production is induced strongly with the fucoidan from bladderwrack and gagome, but with the other kinds there is only a weak effect, as in fig. 3. They assumed that this is probably because the strength of the induction effect differs depending on the structure of the fucoidan. -
Induction of hepatocyte growth factor production
Hepatocyte growth factor (HGF) is a cytokine that was discovered by Nakamura of Osaka University in 1984 in the serum of partially hepatectomized rats and found in 1986 by Goda et al. of Kagoshima University in the serum of a patient with fulminant hepatitis, and the substance is a protein with a molecular weight of approximately 100,000. HGF has extensive physiological effects and is used, for example, in the treatment of cirrhosis of the liver and alcoholic hepatitis as well as for treating ischemic disease caused by diabetes. In 1999 they discovered that F-fucoidan and G-fucoidan promote HGF production. According to Sakai et al. (6) moreover, a comparison of fucoidans from various seaweeds along with heparin with regard to the HGF induction capacity of sulfated fucan yielded the results - HGF induction activity was found in wakame, mozuku, and Lessonia. The 7-sugar 12-sulfate main oligosaccharide obtained when they degraded sulfated fucans derived from gagome with a sulfated fucan-degrading enzyme operated to greatly increase HGF production, nearly to the same degree as heparin. The structure and HGF induction activity of that main oligosaccharide are shown in fig.3. Please have a look at our fucoidan products.
For reference:
- 高橋政壽 1983 日本網内系学会会誌 22, 4, 269-283.
- 丸山弘子ら 2000. 日本栄養・食糧学会総会講演要旨集 54, 126
- H. Sagawa et al. 1999 生化学 71, 203
- 加藤郁之進ら 2004. 海藻の抗癌作用 p.477-490.
- F. Yu et al. 1996. Abstract of X V III th Jap. Carbohydr. Symp., p.93-94
- 酒井武 2003. Jpn.J.Phycol., (Sorui) 51, 19-25
Check out similar products here


